Biotransformation
Definition:
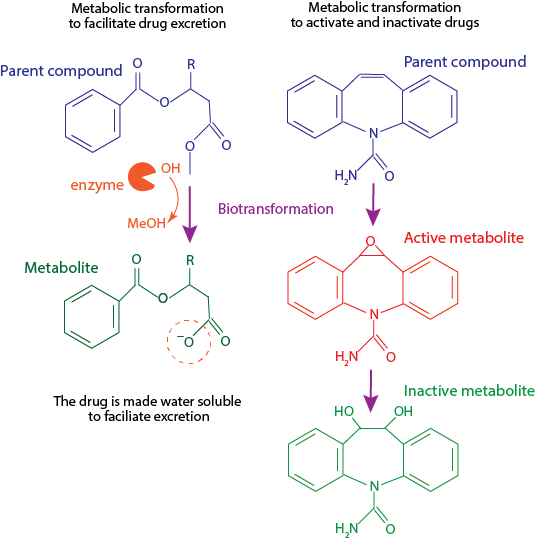
The chemical transformation of a drug to another chemical (often referred to as drug metabolite) in a biological system. This process is also called drug metabolism and represents the M in ADME (absorption, distribution, metabolism, and excretion).
Relevance:
Biotransformation reactions are fundamental to transform endogenous and exogenous molecules into active or inactive and more water-soluble chemicals. Biochemical reactions mediate this process, often referred to as Phase I, Phase II and Phase III (transport-mediated elimination of drugs) reactions. Some of the key biochemical reactions that mediate drug metabolism include oxidation, reduction, hydrolysis and conjugation. These reactions are performed by several enzymes, some of which are prone to genetic polymorphisms, enzymatic induction, and inhibition by other drugs.
Examples of biotransformation:
Into active drug:
- Tamoxifen is metabolized to 4-hydroxytamoxifen, which is 30 to 100-fold more active than its parent drug.
- Codeine is metabolized to morphine, an active compound. Morphine is then further metabolized to morphine-6-glucoronide which has potent analgesic actions.
Into inactive drug:
- Felodipine is extensively metabolized into metabolites with no therapeutic effects.
Into toxic metabolites:
- Acetaminophen is metabolized into a highly reactive intermediate metabolite that is hepatotoxic and nephrotoxic. Under normal conditions, this metabolite is further biotransformed to a nontoxic conjugate, and cleared via bile. However, if overdose occurs, the detoxification pathways become overwhelmed, leading to liver and kidney exposure, and damage by the toxic metabolite.
Linked terms: metabolism, parent compound
Return to Glossary